Zinc
| Zinc | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General Info | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Symbol | Atomic symbol::Zn | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Number | Atomic number::30 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Weight | Atomic weight::65.4 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical series | transition metals | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | bluish pale grey 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group, Period, Block | 12, 4, d | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Ar] 4s2 3d10 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 18, 2 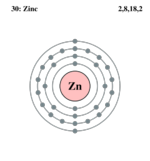
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number | CAS number::7440-66-6 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase | solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density | Density::7.14 g/ml | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | Melting point::692.68 K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | Boiling point::1180 K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of Zinc | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All properties are for STP unless otherwise stated. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
Zinc is a chemical element known by the atomic symbol Zn. It is a widely used bluish white or bluish gray metal. When looking for it on the periodic table, it is located in the fourth period in family IIB. It is somewhat reactive, more so than iron or copper. It is mostly dense, soft, and a semiconductor of electricity. Zinc is able to become brittle, crystalline, ductile, malleable, melted, or boiled, all at different temperatures. Someone might find zinc in rock, soil, air, or water. The human body needs zinc to function. It is rare, but people can die from extremely low zinc deficiency (more on zinc deficiency down below in the video). It can be taken by mouth or intravenously, however regular doses can be harmful.
Properties
Zinc is a lustrous metal that has a bluish-white color[1]. It is shiny, however, if someone was to come across it today, it looks quite dull. It is moderately dense, soft, and a fair conductor of electricity[2]. It is located in group IIB and period four of the periodic table. At room temperature it becomes brittle and crystalline[1]. At temperatures between one hundred and ten degrees Celsius and one hundred and fifty degrees Celsius, it becomes ductile and malleable. At four hundred and twenty degrees Celsius zinc melts, and boils at nine hundred degrees Celsius[2]. Zinc is a fairly reactive metal that will combine with oxygen and other non-metal elements. It will also react with dilute acids to release hydrogen. Zinc is a very common substance that occurs naturally[1].
After iron, aluminum, and copper, zinc is the fourth most widely used metal in the world. Every year, about ten million tons of zinc are produced and used. It is more reactive than iron or copper. When zinc is used with products, it has to be coated first. The reason is because when zinc is exposed to the environment, it develops a tough layer of zinc oxide and zinc carbonate that prevents further corrosion. So when zinc is coated, it protects objects like bridges, fences, lamp posts, or car bodies[2]. Also, the properties of zinc are capable of reducing post-ischemic injury to a variety of tissues and organs. This can be done because zinc is needed in the human body for proper growth. It is also needed for the immune system to function, wounds to heal, blood clotting, thyroid function, and much more[3].
Occurrences
Zinc is a natural component of the earth's crust and a fundamental part of our environment. It is not only present in rock and soil, but also in air, water, and the biosphere. Plants, animals, and humans also contain zinc. Minerals and metals are mostly obtained from the earth's crust. The average natural level of zinc in the earth's crust is seventy mg/kg, however it can range between ten and three hundred mg/kg. Some areas like the earth's surface and underground have much higher levels of concentration by natural geological and geochemical processes. Sphalerite is the most commonly found zinc mineral[4]. It is found in almost all mined zinc deposits (a white powder used as a pigment in paints, cosmetics, glass, and printing inks)[5]. Also, zinc deposits close to the earth's surface are often converted to oxides and carbonates[4]. Native zinc properties have been reported in Australia, New Zealand, and the United States. They have also recently been reported from China and Peru[6]. The largest producers of this mineral are Canada and Japan[7].
Nutrient Deficiencies
Zinc is an essential trace element in the human body. It it found in high concentration in red blood cells as an essential part of the enzyme carbonic anhydrase. This promotes many reactions relating to carbon dioxide metabolism. Zinc is present in the pancreas and may aid in the storage of insulin. In animals, in this case a slug, zinc functions in the hemosycotypsin of snails' blood to transport oxygen. This is similar to iron in the hemoglobin of human blood[6]. Zinc is an element that is vital and essential for human health. Although, if humans take high levels of zinc, it can damage the pancreas, disturb the protein metabolism, and cause arteriosclerosis[1].
People with zinc deficiency can take a zinc capsule to prevent and treat low levels of zinc. Having diarrhea, situations of the bowel not being able to absorb food, and liver cirrhosis are signs of zinc deficiency. If someone is an alcoholic, have had major surgery, or have had a long-term use of a feeding tube in the hospital, are situations that may result in zinc deficiency as well. By taking zinc through the mouth or an IV, it helps restore zinc levels to the appropriate level. However, it is not recommended to take a zinc supplement regularly[3].
In countries that have poverty or that are not well developed, kids are often malnourished. They and most likely the adults as well, probably have severe zinc deficiency. Zinc is likely effective for reducing diarrhea for these children and to help with their low zinc levels. Zinc is also likely effective for treating Wilson's disease[3]. Wilson's disease or hepatolenticular degeneration, is a rare inherited disorder of copper metabolism; copper accumulates in the liver and then in the red blood cells and brain[8]. Taking zinc is possibly effective for treating acne or weak bones. Taking zinc by mouth or applying it to the skin in an ointment containing erythromycin may help clear up acne. For weak bones, or osteoporosis, low zinc intake may be linked to lower bone mass. There are multiple other things that zinc may possibly be effective for, however, need more testing[3].
The Effects of Zinc on the Environment
The world’s zinc production is still rising. This basically means that more and more zinc ends up in the environment. Water is polluted with zinc, due to the presence of large quantities of zinc in waste-water of industrial plants. This waste-water is not purified. One of the consequences is that rivers are depositing zinc-polluted muck on their banks. Zinc may also increase the acidity of waters. Some fish can accumulate zinc in their bodies when they live in zinc-contained waterways. When zinc enters their bodies, it is able to bio magnify up the food chain[9].
Large quantities of zinc can be found in soils. When the soils of farmland are polluted with zinc, animals will absorb concentrations that are damaging to their health. Water-soluble zinc that is located in soils can contaminate groundwater. Zinc cannot only be a threat to cattle, but also to plant species. Plants often have a zinc uptake that their systems cannot handle due to the accumulation of zinc in soils. On zinc-rich soils, only a limited number of plants has a chance of survival. That is why there is not much plant diversity near zinc-disposing factories. Due to the effects upon plants, zinc is a serious threat to the productions of farmlands. Zinc can also interrupt the activity in soils, as it negatively influences the activity of microorganisms and earthworms. The breakdown of organic matter may seriously slow down because of this[9].
Video
A Dr. Joseph Mercola talks about zinc deficiencies and how zinc plays a crucial role in supporting the immune system.
References
- ↑ 1.0 1.1 1.2 1.3 Zinc-Zn LennTech. Web. Accessed October 23, 2013.
- ↑ 2.0 2.1 2.2 Metal Matters: Zinc. ORACLE Think Quest. Web. Last Updated March 20, 2009.
- ↑ 3.0 3.1 3.2 3.3 Find a Vitamin or Supplement: ZINC WebMD. Web. Accessed October 9, 2013.
- ↑ 4.0 4.1 Zinc - Natural Occurrence. International Zinc Association. Web. Accessed October 9, 2013.
- ↑ Zinc Definition. Reverso Dictionary. Web. Accessed October 9, 2013.
- ↑ 6.0 6.1 Zinc (Zn). Encyclopedia Britannica. Web. Accessed October 9, 2013.
- ↑ Occurrence. Nautilus. Web. Accessed October 9, 2013.
- ↑ WordNet Search. WordNet Web. Web. Accessed October 9, 2013.
- ↑ 9.0 9.1 Zinc in the Environment Zinc. Web. Accessed October 23, 2013.
| ||||||||||||||
| H | He | ||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | ||||||||||
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Uut | Fl | Uup | Lv | Uus | Uuo | ||||||||||
|
|||||||||||||||||||||||||||||||||||||||||


