Argon
| Argon | |||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||
| General Info | |||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Symbol | Atomic symbol::Ar | ||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Number | Atomic number::18 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic Weight | Atomic weight::39.948 g/mol | ||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical series | Noble gases | ||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | Colorless; emits blue-violet light in glow discharge 
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Group, Period, Block | 18, 3, p | ||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Ne] 3s2 3p6 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2,8,8 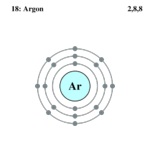
| ||||||||||||||||||||||||||||||||||||||||||||||||
| CAS number | CAS number::7440-37-1 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||||||||||||||||||||
| Phase | gas | ||||||||||||||||||||||||||||||||||||||||||||||||
| Density | Density::0.001784 g/ml | ||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | Melting point::83.80 K | ||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | Boiling point::87.30 K | ||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of Argon | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| All properties are for STP unless otherwise stated. | |||||||||||||||||||||||||||||||||||||||||||||||||
Argon is a chemical element known by the chemical symbol Ar. It is one of the most abundant elements, and the third most common gas in the Earth’s atmosphere. Argon is located in the group VIIIA that is also called noble gases. Noble gases are generally colorless, odorless, and tasteless. Noble gases have extremely low boiling and freezing points. Argon was discovered by a Scottish chemist named Sir William Ramsay, and an English chemist named Lord Rayleigh in 1894. It was isolated by examination of remains obtained by remove nitrogen, oxygen, carbon dioxide, and water from clean air. They named this element “Argon” from Greek word “argos” meaning inactive or indolence, because this gas does not react with anything. The original symbol for Argon was the letter “A” ,then IUPAC changed the symbol to “Ar” in 1957[1].
Properties
Argon is the eighteenth element of the periodic table. It means that Argon has eighteen electrons, eighteen protons, and two neutrons. The atomic number of Argon is 39.948[2].Argon is a colorless, gaseous element which is in the noble gases group and nonmetal group[1]. Argon also has melting point of 83.8 Kelvin (-189.35 Celsius or -308.83 Fahrenheit) and boiling point of 87.3 Kelvin (-185.5 Celsius or -302.53 Fahrenheit)[2], and a density of 1.7837 gram per liter. It has crustal abundance number of 3.5 milligrams per kilogram and oceanic abundance number of 0.45 milligrams per liter (Argon forms an anthracite with “b” hydroquinone which is false chemical bonds). Argon is two and a half times more soluble in water than nitrogen. When you view it through the spectroscope, you will see a lot of red lines[3].
Occurrences
Argon can be found in the Earth's atmosphere, which contains 0.94% of Argon, and Mars' atmosphere contains 1.6% of Argon. Argon-39 is made by cosmic ray activity. There are 22 known isotopes of Argon from Ar-31 to Ar-53 (except Ar-52) [4]. Natural argon is a fusion of three major isotopes: Ar-36 (0.34%), Ar-38 (0.06%), and Ar-40 (99.6%). Not all isotopes of argon have half life, because Ar-36, Ar-38, and Ar-40 are stable, and they will not change their shapes. For example: Ar-37 has 35 days of half life, Ar-39 has 269 days of half life time, and Ar-41 has 1.8 hour of half life[5]. Due to its common nature, the cost of pure Argon of 100 gram is 5 cents[1].
Uses
Argon is used mainly in electrical lights, vivid tubes, photo tubes, glow tubes, and lasers. Argon is used in lighting where nitrogen is unsuitable. It is very important for the manufacture of stainless steels and silicon crystals, because Argon is used as a shield in arc fusing and cutting. Argon-39 is used primarily for applications such as ice coring. A mixture of Argon and Carbon dioxide is sometimes used in metal insert gas fusing of common structures. Argon is also used in wine-making to separate oxygen in barrels[6].
Health
Argon is one of the most health-effecting elements, but Argon does not damage the environment. Argon is used in cryosurgery because it is needed for use in extremely cold temperature to specifically destroy the small areas of diseases under the skin. If one breathes pure Argon in, it will cause sensation of dizziness, headache, suffocation, and eventually death. If one's skin or eyes have contact with liquid Argon, they will become frostbitten[7].
References
- ↑ 1.0 1.1 1.2 Unknown Author. Argon. www.chemicool.com. Web. Access 27 November 2012 .
- ↑ 2.0 2.1 Gagnon, Steve. The Element Argon. education.jlab.org. Web. Access 27 November 2012 .
- ↑ Winter, Mark. Argon: the essentials. www.webelements.com. Web. Access 27 November 2012 .
- ↑ Helmenstine, Anne. Argon Facts. www.about.com. Web. Access 27 November 2012 .
- ↑ Bentor, Tinon. Periodic Table: Argon. www.chemicalelements.com. Web. Access 27 November 2012 .
- ↑ Unknown Author. Argon (Ar) Properties, Uses, Applications Argon Gas and Liquid Argon. www.uigi.com. Web. Access 27 November 2012 .
- ↑ Unknown Author. Argon Ar. www.lenntech.com. Web. Access 27 November 2012 .
| ||||||||||||||
| H | He | ||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | ||||||||||
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Uut | Fl | Uup | Lv | Uus | Uuo | ||||||||||
|
|||||||||||||||||||||||||||||||||||||||||


