Polyvinyl chloride
| Polyvinyl chloride | |
|---|---|
| |
| General | |
| Systematic name | polychloroethene |
| Other names | chloroethene, chloroethylene, PVC |
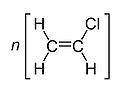
| Molecular formula | (CH2CHCl)n |
| Molar mass | Molar mass::62.498 g/mol |
| Appearance | white powder, most physical properties can be altered with additives |
| CAS number | CAS number::9002-86-2 |
| Properties | |
| Density and phase | [[Density::1.38 g/cm3]], solid |
| Melting point | Melting point::100°C - Melting point::260°C |
| Boiling point | decomposes |
| Hazards | |
| MSDS | Material safety data sheet |
| Main hazards | Can produce harmful gases if burned |
| NFPA 704 | |
| Flash point | 388°C |
| RTECS number | 0.77 |
| Related compounds | |
| Related compounds |
ethylene vinyl acetate |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Disclaimer and references | |
Polyvinyl chloride, or PVC, is made of a long repeating chain of vinyl chloride molecules. During its production, many additives can be mixed into the substance to change its physical and chemical properties.[1] Because of this, it is a very versatile material which can be used for a variety of purposes. Many objects in today's world, such as pipes, roof shingles, toys, and wire sheaths are made with PVC. PVC does have some potentially negative side effects, though, so many groups are trying to have people stop using PVC.[2]
Properties
Polyvinyl chloride is a very versatile substance, and it has many different physical and chemical properties. It is very hard and rigid, and does not easily bend once it has hardened. This means that unless certain additives are used, it is typically brittle, and tends to splinter under excess pressure. It is also fairly light, with a molar mass of only 62.498 grams per mole. Its lightness and great strength makes it very useful in construction. It is also a very durable substance; most acids and bases do not affect it, although some solvents and chlorinated hydrocarbons may damage PVC. Its melting point is fairly low, so it cannot be used for hot water pipes. It is also resistant to electricity, making it ideal for use in the covering around electrical wires. Its color can vary depending on the additives. PVC is also not biodegradable. This means that it will not naturally break down into other substances over the course of time.[3]
Polyvinyl chloride is almost always made with extra additives. Compatibilizers, flame retardants, antioxidants, and plasticisers can be added to PVC to change its properties. Plasticisers, for example, make polyvinyl chloride much more flexible while still retaining its strength. Other additives can make PVC more resistant to flame and chemicals. Its color can also be changed by adding different pigments. Inert materials called fillers can also be added which increase the amount of PVC being made. Compatibilizers make the PVC more compatible with other substances that are also added.[1]
Occurrences
Polyvinyl chloride itself does not exist naturally. Instead, it is made in large factories through a relatively cheap and simple process. First, ethylene and chlorine are combined. This makes a product called ethylene dichloride. This is then turned into vinyl chloride, the base molecular unit of polyvinyl chloride. Through a process called polymerization, the vinyl chloride forms into long chains of polyvinyl chloride. PVC in this form is a white powder. Additives, dyes, and plasticisers can be added at this point which change the properties of the PVC so it can be used for a variety of different purposes.[4]
Uses
Polyvinyl chloride is probably one of the most ubiquitous substances in today's world. You are probably sitting on an object containing PVC right now. Because of its great strength and durability, PVC is used for many different purposes. Many children's toys and baby products are made from PVC, as they need to be durable so the child will not break it easily. Vinyl records were made from PVC. Credit cards, imitation leather, and plastic packaging are also made from this versatile substance. Typically, water bottles are also constructed from polyvinyl chloride, but many environmentalists are trying to change this because the water bottles clog up landfills. Polyvinyl chloride is typically used when building houses. Roof tiles, window frames, gutters, floors, insulation, siding, and some doors can be made with PVC. Many clothing items contain PVC. Rubber boots, shoes, aprons, and t-shirts can contain or be made with this substance.[5]
PVC is used widely in construction. In fact, nearly seventy percent of the plastics used in construction are products of polyvinyl chloride.[6] Generally, PVC is used in making pipes for plumbing. PVC is durable, strong, and can be welded to other objects to make a perfect seal, making it ideal for this purpose. Most of the pipes made of PVC are used for waste disposal because polyvinyl chloride does not allow any of the waste products in the pipe to leak out into the surrounding ground.[5]
Polyvinyl chloride is also widely used in the automotive industry. PVC is nearly as strong as metal but is both cheaper and easier to manufacture, so some metal parts in cars can be replaced with plastic ones. The dashboard, lining, and parts of car seats are typically made with PVC. The door panels, sound insulators, and protection for the underside of the car can also be made with polyvinyl chloride.[7]
Polyvinyl chloride is also used in wiring. PVC is an excellent insulator, and it is very flexible, so it is perfect for use as a wire sheath. PVC covers the wires in devices such as telephones, computers, televisions, data cables, speakers, computer monitors, washing machines and many other electrical appliances. While PVC is mainly found in the wiring of these devices, it can also be used in making the circuit cards and component capsules.[5]
Health Hazards
During the production cycle of polyvinyl chloride, a chemical called dioxin is produced. Dioxin is very toxic, and can lead to many health problems such as cancer, birth defects, and neurological damage. Dioxin is chemically stable, so it does not break down. Instead, it is consumed by animals and humans, and accumulates in the fatty tissue, potentially causing many health issues. Dioxin is not the only harmful chemical involved in the PVC production process. Many substances are used during production to stabilize the process. These can include lead and cadmium, which can later possibly leech out of the PVC. Phthalate plasticisers are also added to PVC while it is being made, and these can harm humans if they are ingested. PVC is sometimes used in children's toys, and some groups claim that if a child were to put a toy constructed of PVC into their mouth, they could ingest small amounts of harmful substances like lead and phthalates.[2]
While polyvinyl chloride is flame retardant, it does release harmful gases when it is burned. It releases hydrogen chloride long before it starts to burn. This gas, when inhaled, turns into hydrochloric acid and can seriously damage a person's lungs and throat. Dioxin, the harmful toxin released during production, can also be released when PVC is burned. Since PVC is commonly used as the sheath around wires, dioxin and hydrogen chloride can be released in large quantities during building fires when all of the electrical wires in the walls burn.[2]
PVC does not biodegrade easily. While this makes it ideal for use in making pipes, this means that PVC can quickly fill up landfills as it will not decompose. While some polyvinyl chloride can be recycled, the large amount of additives used in PVC makes it difficult to recycle. Only about one half of one percent of the PVC that is thrown away is recycled.[2]
Because of all of the above reasons, many groups, such as Greenpeace, are working to replace PVC with healthier and greener alternatives. Many companies have also stopped using polyvinyl chloride in their stores and products. Even governments have started to take action. Both Swedish and Danish governments have restricted the production of PVC.[8]
References
- ↑ 1.0 1.1 Author Unknown. [1] lenntech.com. Web. Accessed January 29, 2012
- ↑ 2.0 2.1 2.2 2.3 Author Unknown. [2] Healthybuilding.net. Web. Accessed January 29, 2012
- ↑ Author Unknown. [3] EngineeringToolbox.com. Web. Accessed January 29, 2012
- ↑ Author unknown. [4] pvc.org. Web. Accessed 25 January 2012
- ↑ 5.0 5.1 5.2 Author Unknown. [5] Greenpeace.org. Web. Accessed January 29, 2012
- ↑ Author Unknown. [6] Hultec. Web. Accessed January 29, 2012
- ↑ Author Unknown. [7] Rusvinyl.ru. Web. Accessed January 29, 2012
- ↑ Author Unknown. [8] Greenpeace.org. Web. Accessed January 29, 2012
| ||||||||||||||




